Category Archives: Documentation
Question Regarding PIC/S PE009-14
The current GMPs in Australia adhere to the PIC/S Guide to Good Manufacturing Practice, (PE009-13). This guide was adopted by the TGA in 2018 and the TGA has “just given” notice that it intends to adopt the current version of the PIC/S Guide (PE009-14) by July 1st 2020.
The Question posed on LinkedIn:
“…I have read PIC/S guidelines, when I was working in Pharmaceutical company. Currently, I am not working in Pharmaceutical company. I would like to keep my knowledge updated about what’s new in regulatory guidelines in Pharmaceutical. Would you please let me know, where can I get copy of new PIC/S??…“
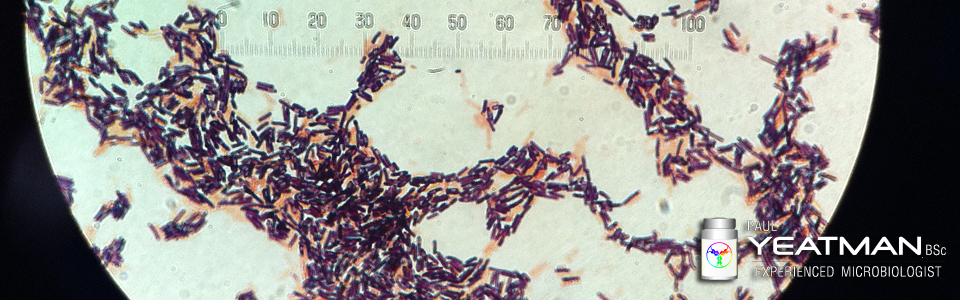
Question Regarding Making Cephalosporin and Cytotoxic Drugs in the Same Building
The Question posed on LinkedIn:
“Dear Experts: Is it possible to make Cephalosporin and Cytotoxic drug in a same building considering Risk Assessment ?“

Book Review of “Eliminating the Gobbledygook” by Kathy Walsh
As one with a love for documentation and trying to communicate effectively and concisely, I bought a copy of Eliminating the Gobbledygook – Secrets to Writing Plain Language Procedures by Kathy Walsh. I am pretty sure I worked alongside Kathy some years back on what was called the Documentation Effectiveness Project at Mayne so I was not expecting too many surprises as we both know our stuff. The purpose of the book purchase was to refresh and update my knowledge of controlled document writing. I figured I may as well review the book in the process. Continue reading

What is Data Trending and How To Identify, Action and Report Trends
The following is an article I placed on LinkedIn and was written as part of my Developing My Writing While Helping Others series.
Paul Yeatman is a microbiologist with over 15 years’ experience in documentation, validation and running investigations in TGA and FDA regulated environments. He has a strong interest in process improvement, documentation, training and developing others. From 9-5 Paul investigates and solves software problems. By night he works on his science chops. He has an arty streak, runs several blogs and enjoys communicating his experiences and knowledge in arenas such as this. Continue reading
Question regarding Bacillus subtilis recovery when testing for absence of Salmonella sp.
The Question posed on LinkedIn:
“Dear experts, During microbial contamination testing for specific organisms – salmonella growing was observed. identification shows that is not Salmonella, but Bacillus subtilis. How i shoud interprated the results, pass specification or not?“
Question regarding sterile filter hold time validation
The Question posed on LinkedIn:
“How to validate maximum hold time for product sterile filter after autoclave sterilization? Any link to regulatory guidelines is much appreciated.“

Controlled Documentation. Identifying what goes into a Policy, a SOP and an OI
The following is an article I placed on LinkedIn and was written as part of my Developing My Writing While Helping Others series.
I am a microbiologist with over 15 years’ experience in the pharmaceutical realm. I have a strong interest in regulatory compliance, documentation and developing others. Recently I have been working closely with data security. I have an arty streak, have developed and delivered training and have an affinity for computers. I ride bicycles…a lot.
Controlled Documentation. Identifying what goes into a Policy, a SOP and an OI Continue reading