Introduction
I wrote the first version of this article in 2014 based on training I developed in 2006. I have updated and reviewed the content to ensure it remains relevant in 2020.
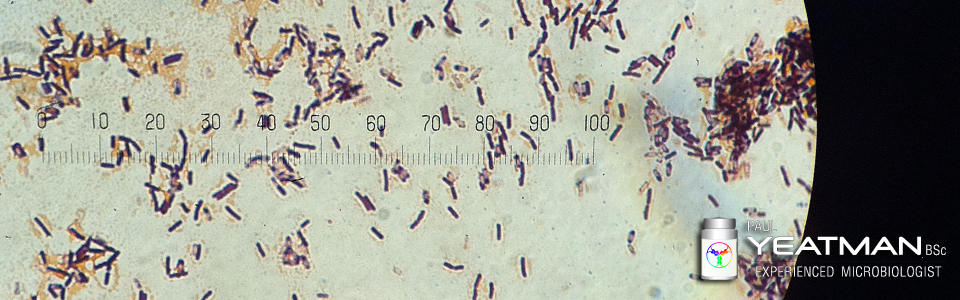
When testing non sterile products, as well as allowed numbers of microbes, there are certain types of microbes that are specified in the regulations as not allowed and still others that can de deemed to be objectionable.
If you work as a microbiologist where you test total microbial aerobic counts (TMAC), total yeast and mould counts (TMYC) and for specific microbes or conduct investigations into the nature of microbes and what impact they may have on the product and the end user, this presentation will benefit you. I will teach you the the following:
- What makes a specified organism
- What makes an objectionable organism
- How to determine if a microbe is objectionable
- Risk assessment considerations
- The benefits of knowing the differences between specified & objectionable organisms