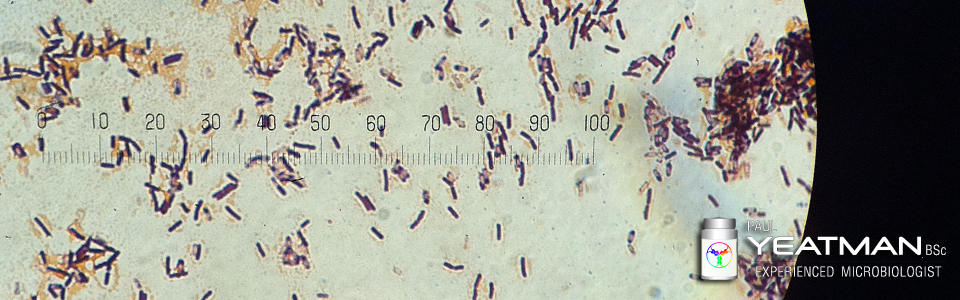
The TMC is broken into two parts: a basic look for contamination and a look for more specific forms of contamination. The basic test is described in the USP in section <61> or in the EP in section 2.6.12. The more specific or specified microbe testing is described in USP section <62> or in the EP in section 2.6.13.
Basic total viable microbial count test:
This tests the product or raw material for a general range of bacteria and yeasts/moulds using general media and test methods. The test gives an overall indication of the state of microbial contamination of the test sample.
Test for specified microbes:
Testing for these microbes is based on objectionable food industry microbes (refer to the FDA bad bug book, 2nd ed 2012). In reality, specified microbes may or may not cause a problem when detected in a sample. However, the regulations require that they not be present.
As discussed in another post, What Makes An Objectionable Organism?, how the objectionability (sic) of all microbes recovered during TMC testing needs to be evaluated.
What is TYMC?
When Sabouraud Dextrose (SDA) plates are used to recover microbes, the media is selecting for yeasts and moulds. Due to this, the obtained is the Total Yeast and Mould Count. This is the case whether or not the method specifically says to test for yeasts and moulds.
What is TAMC?
When Tryptone Soya Agar plates are used to recover microbes, the media is selecting for bacteria. Due to this, the result obtained is the Total Aerobic Microbial Count. Sometimes this is called the Total Bacteria Count or TBC.
What is TMC?
The Total Microbial Count (also known as the Aerobic Microbial Count) is the sum of the TYMC and TAMC counts. i.e. TMC = TYMC + TAMC.
Why are Certain Media, Incubation Temperatures and Incubation Periods used?
Where a product is specifically listed in a pharmacopeia, it is said to have a monograph. This contains all the required testing for a product – one of these tests should be the TMC test. If this is not the case, technically the product does not need to be tested, but it really should be. This goes beyond best practice and indicates to an auditor that you do not just do the bare minimum to get a product out the door.
Regardless of the presence of a monograph, the section of the pharmacopeia that deals with the test specified the media, incubation temperatures and times. Links to these have been included for you reference.
Why is validation necessary?
Generally, one only needs to show that a method is equivalent to the regulated method. This is not always possible, as a product may contain substances that are inhibitory to microbes. A good example is products which contain antibiotics or inhibitory factors.
The purpose of validation is to show that if a range of common microbes are present within the product (for the USP 61 test) or if any of the specified microbes are present (USP 62), your test method will recover them.
In simple terms, a validation is conducted by adding a known number of microbes to various test samples and after plating the sample and incubating them the recovery counts are examined to determine if the microbes can be recovered. Typically, a not less than 75% rule is applied here. As well as specified test microbes, if you have a few microbes that consistently are recovered from your Viable Environmental monitoring program, it is a good idea to add them to your list of validation microbes.
Where you cannot show recovery, your should still test the products as microbes not used during your validation may grown in your product and cause contamination issues. Not being able to recover microbes from a product during validation is helpful when conducting investigations into product contamination. If your validation showed you could not recover microbe A, but an out of specification result indicated the product was loaded with microbe A, part of your investigation should discuss this. This could mean your test plates were contaminated. It could also mean you have a have a REALLY big contamination issues as the antimicrobial substances in your product were out gunned by the sheer number of microbes. As for any investigation, the conclusions you drawn must be supported by evidence.
Why is it necessary to test for TMC? (What may happen to your customer)
So, besides it being a regulatory requirement, why do we bother testing for TMC?
You want to ensure that what you are providing to your customers is a product that is what you purport it to be and it will do what you say it will do. A product containing a large number of microbes may not be potent or it may cause disease or death. Eg:
- A contaminated oral suspension may cause ulcers or a gastro intestinal (GIT) upset.
- A contaminated skin cream may cause a localised skin infection
- A contaminated anti fungal cream may cause a bacterial infection and not cure the yeast infection
- A contaminated tablet may cause a GIT upset
- A contaminated injectable product may cause septicaemia or even block a capillary if contaminating particles were large enough
- A contaminated injectable product may also cause a fever in a patient.
As well as this, certain microbes are capable of inactivating antibiotics. A good example is gram negative rods. These can inactivate gramicidin.
How should results be recorded?
For the generalised testing, counts are recorded as “<10CFU/weight” event when a zero count is obtained. If you validate your limit of detection, this figure could change. As it stands, the 10CFU/weight is a guideline. This also means that any result obtained that is less than 10 is recorded as less than 10, even if the result is 2 or 6 or 9 etc. Depending on the class of product you are testing, you may want to identify any microbes you have recovered (good practice and it also helps in determining of the microbe is objectionable). For specified microbes, a testing lab conducts tests that specifically look for the microbe. If one of these tests does not reveal any of the specified microbes, there is a high chance that the microbes will not be recovered during TAMC/TYMC testing.
For specified microbe testing, the result is as present (a pass) or absent (a fail).
Appendix:
Did you find this informative or useful? Please consider a small donation so I can expand and improve on what I deliver.
![]()